 
The name mustard gas is technically incorrect: the substances, when dispersed, are often not gases but a fine mist of liquid droplets. Also known as mustard agents, this family of compounds are infamous cytotoxins and blister agents with a long history of use as chemical weapons. Such compounds are potent alkylating agents, which can interfere with several biological processes. 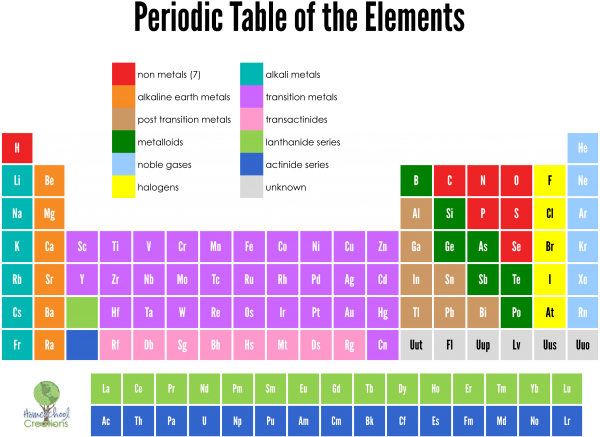
In the wider sense, compounds with the substituent S(CH 2CH 2X) 2 and N(CH 2CH 2X) 3 are known as sulfur mustards and nitrogen mustards, respectively, where X = Cl or Br. Ideal for use by teachers, educationists, scientists, laboratory physicians, etc.Mustard gas or sulfur mustard is any of several chemical compounds that contain the chemical structure S(CH 2CH 2Cl) 2. Also, this template will be useful for university teachers when preparing materials in chemistry.ġ00% editable and customizable periodic table template where you can change color and text to meet your preference.ĭownload the Periodic table template and use it to make stunning scientific and chemistry presentation. You can use this template to prepare information about chemical experiments or new materials to help replace organic materials with inorganic ones. This template will be useful for employees of scientific institutes specializing in the development for the chemical industry. This template allows you put together a fast and easy PowerPoint presentation on periodic table without any need for graphic skills. Run comparison between two different groups in the period table with the fourth slide. The user can make use of the third slide to give a snapshot of a group and all the element in it while using one as a topic of discussion. Features 10 legends for explanation of the color codes. The periodic table in the first and second slides are categorically color-coded according to their group. 
The period table template provides information about the elements, their groups, and the various trends they show. Periodic table PowerPoint Template is a presentation design template containing the number of elements in the periodic table. Moving down a group, elements have successively lower ionization energies because it becomes easier to remove an electron from the outermost shell. Electronegativity decreases moving down a group because adding an electron shell pushes the valence electrons further from the nucleus. Atomic radius increases moving down a group, as successive elements gain an electron energy level. Elements within a group display a pattern with respect atomic radius, electronegativity, and ionization energy. Elements with a group share a valence electron configuration. Groups are numbered from 1 (the alkali metals) to 18 (the noble gases). The columns of elements are called groups or families. Moving down a period on the table adds a new electron shell. Elements toward the left side of a period are metals, while those on the right side are nonmetals. Element atomic number increases moving from left to right across a period. There are seven rows of the periodic table, which are called periods. The structure of the periodic table makes it possible to see relationships between elements at a glance and predict properties of unfamiliar, newly discovered, or undiscovered elements. However, his table illustrated recurring trends or periodicity in the element properties. Mendeleev’s table ordered the elements according to increasing atomic weight rather than atomic number. The modern table is derived from Mendeleev’s periodic table, but with one significant different. The Russian scientist Dmitri Mendeleev is most often credited with inventing the periodic table (1869). 
The periodic table is a tabular arrangement of the chemical elements by increasing atomic number which displays the elements so that one may see trends in their properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
